 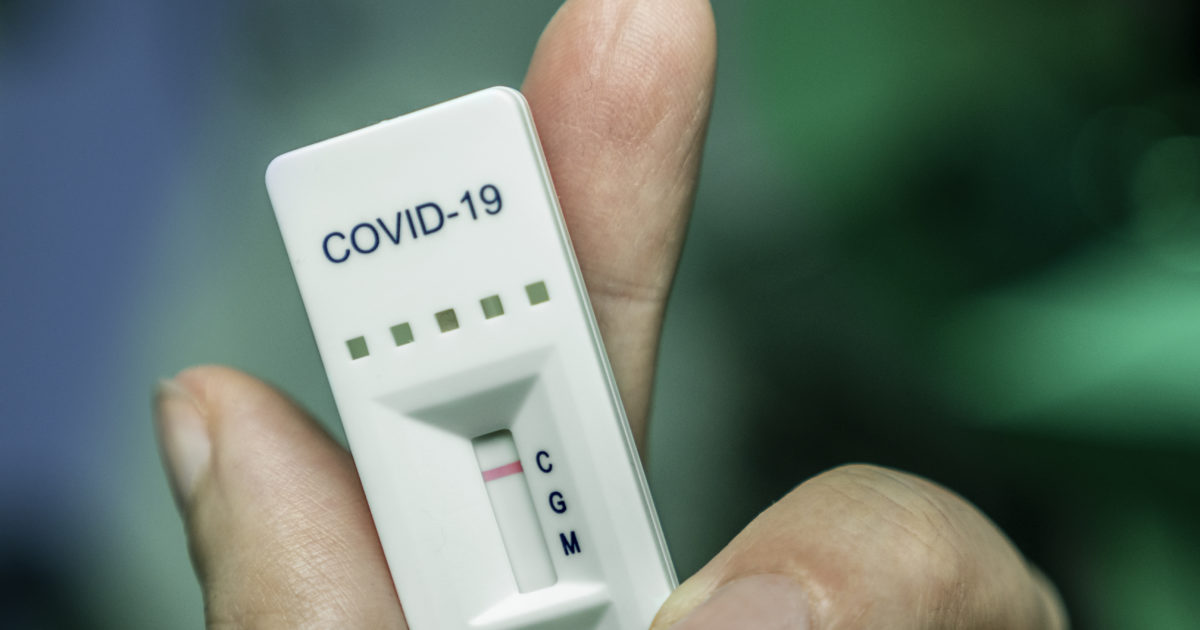
The Assure COVID-19 IgG/IgM rapid test device should not be used to diagnose acute SARS-CoV-2 infection. 
The SARS-CoV-2 genome is composed of two polypeptides encoded between two open-reading frames that are processed by viral proteases to produce nonstructural proteins (). SARS-CoV-2 is an enveloped, single-stranded ribonucleic acid (ssRNA) virus. 
At this time, it is unknown for how long antibodies persist following infection and if the presence of antibodies confers protective immunity. Potential for Multiorgan Involvement in COVID-19. The Assure COVID-19 IgG/IgM rapid test device is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection. Molecular Diagnostics Toenail Fungus Panel (Onychomycosis) BD MAX CT/GC/TV Assay UTI/ABR Analysis BD MAX Vaginal panel COVID-19 Policy. Premier Medical Laboratory Services (PMLS), headquartered in Greenville, South Carolina, is the official health and wellness partner of the Greenville Triumph and an advanced molecular diagnostics. HealthTrackRx is an industry leading infectious disease laboratory providing accurate, targeted molecular diagnostics. The Assure COVID-19 IgG/IgM rapid test device is a rapid lateral flow chromatographic immunoassay intended for the qualitative detection and differentiation of IgM and IgG antibodies to SARS-CoV-2 in human venous whole blood (sodium EDTA), serum, plasma (sodium EDTA) and fingerstick whole blood. Future pandemics are always a possibility, and lessons must be learnt from the current pandemic to enable robust and expedited responses. Premier Biotech FaStep® AZ COVID-19P Rapid Test Device - 20/Box While the more affluent countries have outcompeted the rest on the COVID-19 diagnostics market,17 many developing countries have used what they have at their disposal to scale-up testing. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
